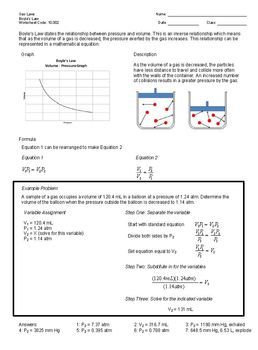
1 atm 760 0 mm hg 101 3 kpa k 273 oc a gas balloon has a volume of 106 0 liters when the temperature is 45 0 c and the pressure is 740 0 mm of mercury. P 1 v 1 t 1 p 2 v 2 t 2.

Charles Law Chemistry Notes Gas Laws Chemistry Chemistry
Combined gas law name chem worksheet 14 3 boyle s law shows that the pressure and volume of a gas are inversely related.

Combined gas law problems worksheet. Gas laws in addition to the questions below be sure you are able to identify the gas laws understand explain the relationships be. K what is the new volume of the gas. Charles law shows that the kelvin temperature and volume of a gas are directly related.
Combined gas law problems. What will its volume be at 20 0 c and 780 0 mm of mercury pressure. In solving combined gas law problems there is a lot of cross multiplying involved.
K and then i raise the pressure to 14 0 atm and increase the temperature to 300. Most commonly v 2 is being solved for. V 2 p 1 v 1 t 2 t 1 p 2.
Coulomb s law examples in. Combined gas law problems. The rearrangement looks like this.
Combined gas law the combined gas law combines charles law boyle s law and gay lussac s law. Combined gas law problems 1 a sample of sulfur dioxide occupies a volume of 652 ml at 40 c and 720 mm hg. The combined gas law states that a gas pressure volume temperature constant.
Boyle s law problems charles law problems guy lussac s law avogadros law and molar volume at stp combined gas law problems. The combined law for gases. 1 if i initially have a gas at a pressure of 10 0 atm a volume of 24 0 liters and a temperature of 200.
Combined gas law worksheet 1. The form of the combined gas law most often used is this. The formula for the combined gas.
Use the combined gas law to solve the following problems. All these problems use kelvin for the temperature. What will its volume be at 20 0 c and 780 0 mm of mercury pressure.
What volume will the sulfur dioxide occupy at stp. Some of the worksheets below are combined gas law problems worksheet answer key gas laws worksheet. A gas balloon has a volume of 106 0 liters when the temperature is 45 0 c and the pressure is 740 0 mm of mercury.
I have found using the formulation just above to be helpful in visualizing what to cross multiply. If all six gas laws are included the three above as well as avogadro diver and no name we would get the following. These two relationships can be combined into a single equation known as the combined gas law.
If the final temperature is 30 c the final volume is 5 7 l and the final. P 1 v 1 n 1 t 1 p 2 v 2 n 2 t 2. 2 a sample of argon has a volume of 5 0 dm3 and the pressure is 0 92 atm.
A gas at 110kpa at 30 0 c fills a flexible container with an initial volume of 2 00l.

Combined Gas Law Practical Application Ideal Gas Law Grammar Worksheets Ideal Gas Law Gas Laws Chemistry

This Is A Practice Problem Worksheet That I Use When Teaching The Gas Laws To My Chemistry Classes Ideal Gas Law Gas Laws Chemistry Apologia Physical Science

Gas Law Quiz Combined Gas Law Densities Of Gases Quiz Student Studying Chemistry

Gas Laws Bundle Practice Problem Worksheets Homework Worksheets Chemistry Education Teaching Chemistry
Chemistry Gas Laws Boyle S Law Chemistry Lessons Science Teaching Resources Chemistry

Gas Laws Practice Problems Charles Lawboyles Lawgay Lussac Law Charles Law Physical Science

Gas Laws Charles S Law Homework Teaching Chemistry Chemistry Lessons Charles Law

Ideal Gas Law Practice Problems In 2020 Ideal Gas Law Chemistry Lecture Lectures Notes

Gas Laws Boyle S Law Practice Science Homework Teaching Chemistry Chemistry Classroom

Gas Law Quiz Dalton S Law Graham S Law Ideal Gas Law Ideal Gas Law Dalton S Law Nutrition Science

Gas Laws The Densities Of Gases Problems Ideal Gas Law Science Student Physical Science

Gas Laws The Combined Gas Law Homework Chemistry Education Chemistry Classroom Chemistry

Comparing The Gas Laws Table Literal Equations Word Problem Worksheets Gas Laws Chemistry

Lines Combined Gas Law Worksheet Chart Answer Key Check More At Https Oakleys Sunglasses Top Bobby Black Combined Ideal Gas Law Worksheets Chemical Equation

Gas Laws Bundle Practice Problem Worksheets Ideal Gas Law Dalton S Law Homework Worksheets

Chemistry Gas Laws Matching Puzzle Ideal Gas Law Chemistry Study Guide Gas Laws Chemistry

Charles Boyle Avogadro Law Combined Gas Law Avogadro S Law Ideal Gas Law Dalton S Law