1 antimony tribromide sbbr3 2 hexaboron silicide b6si 3 chlorine dioxide clo2 4 hydrogen iodide hi 5 iodine pentafluoride if5. Chapters 6 and 7 worksheet spring 2013 page 1 of 5 chapters 6 and 7 practice worksheet.
Ionic And Covalent Bond Worksheets Covalent Bonding Worksheet Covalent Bonding Ionic And Covalent Bonds
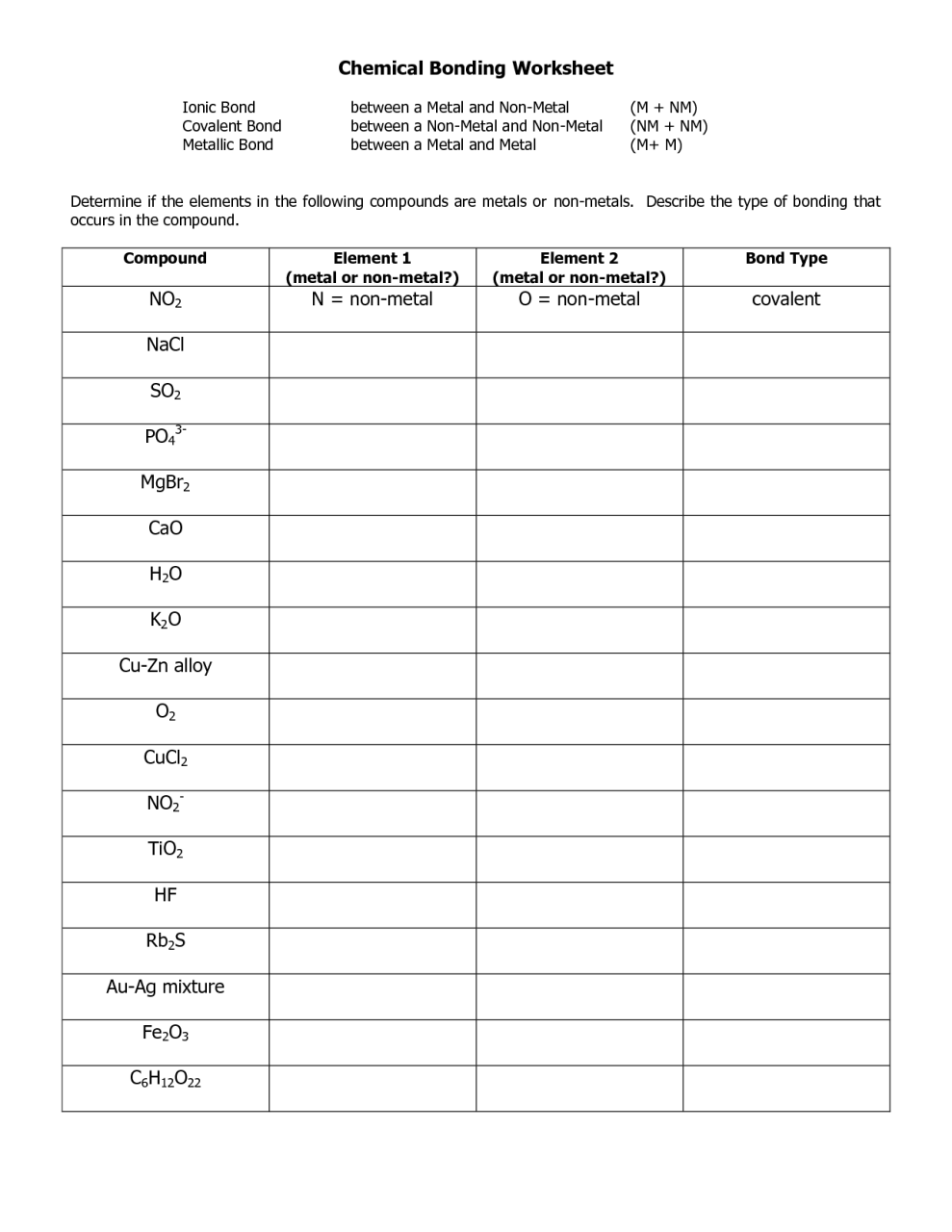
Worksheet chemical bonding ionic and covalent answers part 2 together with covalent bonding worksheet.
Covalent bonding worksheet answers pdf. This is most often between non metal atoms but there are a number of compounds between metals and non metals that are covalent. Write the electron dot structure lewis dot structure for covalent compounds or ions. For example boron atoms bonded to oxygen atoms are covalent.
Unit 4 covalent compounds 1. Ionic bonding is often the outcome of the covalent bond as well. A single covalent bond involves one shared pair of electrons.
Covalent bonding name covalent bonding occurs when two or more nonmetals share electrons. 1 hydrogen hydrogen diatomic element 1 write the symbols for each element. Naming covalent compounds solutions write the formulas for the following covalent compounds.
Covalent compound naming worksheet 1 covalentname sxw. Given the formula of a covalent compound write its correct name. Use electronegativity to determine the polarity of a bond or molecule.
Covalent bonds and molecular structure 1 how are ionic bonds and covalent bonds different. Covalent bonding involves the sharing of electron pairs between two atoms. Showing top 8 worksheets in the category covalent bonding chapter 2.
Attempting to attain a stable octet of electrons at least part of the time. In ionic bonding the atom of the element is bonded to an atom of the opposite sign. Some of the worksheets displayed are chapter 7 practice work covalent bonds and molecular chapters 6 and 7 practice work covalent bonds and chapter 8 covalent bonding and molecular structure 6 chemical bonding bonding basics covalent bonding work chapter 6 05 ctr ch08 71204 812 am 181 molecular compounds 8.
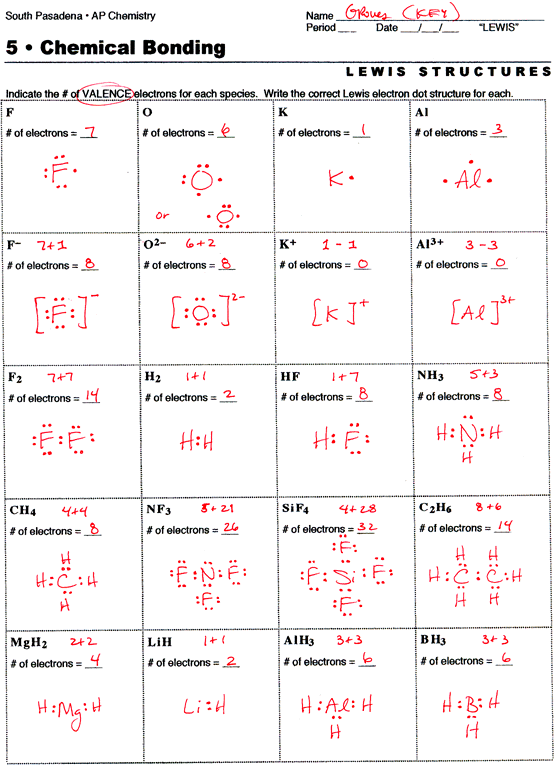
2 describe the relationship between the length of a bond and the strength of that bond. H h sharing the electron pair gives each hydrogen an electron configuration analogous to helium. Given the name of a covalent compound write its formula.
Naming covalent compounds solutions write the formulas for the following covalent compounds. 3 identify the type s of bond s found in the following molecules. Show how covalent bonding occurs in each of the following pairs of atoms.
2 use fruity pebbles or other cereal candy with more. 1 antimony tribromide sbbr 3 2 hexaboron monosilicide b 6si 3 chlorine dioxide clo 2 4 hydrogen monoiodide hi 5 iodine pentafluoride if 5 6 dinitrogen trioxide n 2o 3 7 ammonia nh 3 8 phosphorus triiodide pi 3. H two hydrogen atoms each with 1 electron can share those electrons in a covalent bond.
Bonding basics covalent bonds answer key teacher notes complete the chart for each element. Covalent bonding in h2 h. Follow your teacher s directions to complete each covalent bond.
Note that hydrogen is content with 2 not 8.

Ionic Compounds Names And Formulas Worksheet November 17 2017 Covalent Bonding Worksheet Practices Worksheets Covalent Bonding

Ionic And Covalent Bonding Practice Worksheet Answers Worksheets

This Could Be Used As An Assessment Or Practice Worksheet Over Chemical Bonding Covalent Bonding Worksheet Chemistry Worksheets Covalent Bonding

Chemical Bonding Basics Practice Worksheet Practices Worksheets Covalent Bonding Worksheet Science Today

Igcse Identifying Ionic Covalent Bonds Covalent Bonding Ionic And Covalent Bonds Chemistry Worksheets

Covalent Bonding Practice Worksheet Practices Worksheets Covalent Bonding Physical Science

Ionic Bonds Student Exploration Gizmo Worksheet Ionic Bonding Covalent Bonding Worksheet Covalent Bonding

Ionic Vs Covalent Coloring Activity Chemistry Science Pdf Printable From Laurelsusanstudio On Teacher Chemistry Lessons Teaching Chemistry Chemistry Classroom

Printables Chemical Bonding Worksheet Chemical Bonding Worksheet Covalent Bonding Worksheet Covalent Bonding Chemical Bond

Practice With Covalent Bonding Covalent Bonding Covalent Bonding Worksheet Teaching Chemistry

This Activity Is A Way To Practice Classifying Chemical Formulas As Either Ionic Or Covalent Bonds Covalent Bonding Ionic And Covalent Bonds Teaching Chemistry

Lewis Dot Structure Covalent Bonds Worksheets Chemistry Worksheets Chemistry Exam Study

Covalent Bonding Worksheet Answer Key Beautiful Naming Binary Covalent Pounds With Key In 2020 Naming Compounds Worksheet Grief Worksheets Covalent Bonding

Bonding Worksheet Doc Covalent Bonding Worksheet Covalent Bonding Ionic Bonding

Ionic Bonds Worksheet Chemistry Worksheets Ionic Bonding Ionic And Covalent Bonds

Covalent Bond Covalent Bonding Covalent Bonding Worksheet Math Worksheets

Covalent Bonding Worksheet Covalent Bonding Worksheet Covalent Bonding Ionic Bonding

Chemical Bonding Worksheet Pdf Nidecmege Covalent Bonding Worksheet Answers In 2020 Covalent Bonding Worksheet Covalent Bonding Kids Worksheets Printables


