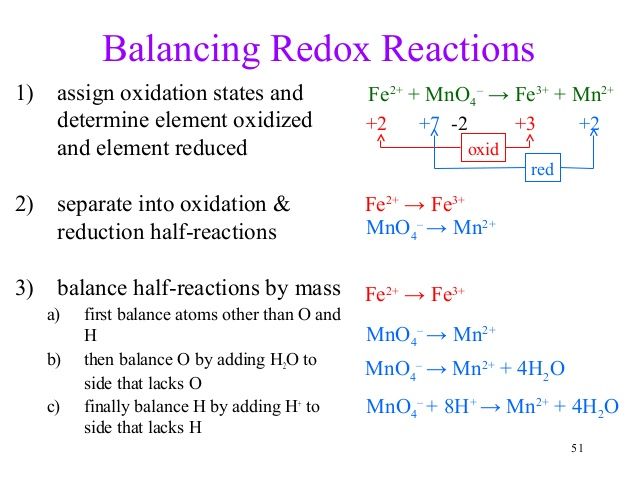
Be sure the reaction is redox look at the oxidation numbers for the atoms in the reaction. In the first case you separate out the oxidation and reduction half reaction and in the second case you do it all at once.
Redox Reaction Redox Reactions Teaching Chemistry Chemistry Notes
Overall scheme for the half reaction method.

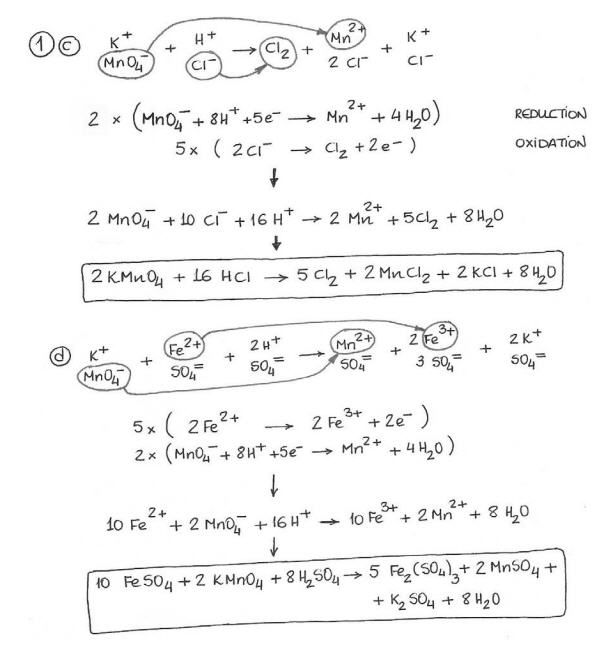
Balancing redox reactions worksheet 1. Balancing redox reactions chem 1a b steps for balancing redox reactions with the reaction method. Redox practice worksheet name. In the reaction al0 cr3 al3 cr0 the reducing agent is a.
Mno 2 mn 2o 3 balance each redox reaction in acid solution using the half reaction method. In the reaction 2k cl2 2kcl the species. Balance the charge or oxidation number with electrons.
The half reaction method and the change in oxidation method. We will use a metal displacement reaction to explain how balancing redox reactions using the oxidation state methods works. Identify the pair of elements undergoing oxidation and reduction by checking oxidation states.
In which substance is the oxidation number of nitrogen zero. Balance each of the following half cell reactions. Split reaction into half reactions reduction and oxidation step 2.
In each case assume that the reaction takes place in an acidic solution also state whether the reaction is oxidation or reduction. Balancing redox reactions worksheet 1 balance each redox reaction in. Al s cuso 4 aq al 2 so 4 3 aq cu s.
Cr 2o 7 2 cr3 5. Mn 2 bio3 æ mno4 bi 3 mno4 s2o3 2 æ s4o6 2 mn 2. Teo 3 2 n 2o 4 te no 3 10.
For example in the oxidation. The two half reactions in table pageindex 1 also illustrate another important feature of balancing redox reactions. What is the oxidation number of carbon in nahco3.
Notice that in each half reaction there is a balance both in the numbers of atoms of each kind and in the overall charge on each side. Considering the equation above we have 2 hydrogen h with the total charge 1 refer the charges of the elements in the above table and 2 oxygen o with the total charge 2 on the l h s and 2 hydrogen h with total charge 2 and only 1 oxygen o with the total charge 2 on the r h s. No no 3 6.
Ws 4 balancing redox reactions. The half equation method separates the oxidation and reduction of a redox reaction in half reactions. H 2o 2 cr 2o 7 2 o 2 cr 3 9.
1 balance the given redox reaction. The oxidation numbers of some elements must increase and others must decrease as reactants go to products. So 4 2 so 2 7.
H 2 o 2 2 h 2 o. Balance o by adding h 2o. They actually involve the same procedure.
Worksheet on balancing redox equations two methods are often mentioned for balancing redox reactions. Worksheet 5 balancing redox reactions in acid and basic solution balance each half reaction in basic solution.

Balancing Chemical Equations Chemical Equation Chemistry Worksheets Balancing Equations

Redox Balancing Redox Reactions Chemistry Lagrange College

49 Balancing Equations Practice Worksheet Answers In 2020 Balancing Equations Chemical Equation Equations

Balancing Equations Worksheet Answers Beautiful As Chemistry Balancing Redox Equations Worksheet In 2020 Balancing Equations Persuasive Writing Prompts Equations

Chemistry Multiple Multiple Choice Types Of Chemical Reactions Chemistry Chemical Reactions Multiple Choice
Pin By Saitech Informatics On Balancing Of Chemical Reaction Equations Distance Time Graphs Worksheets Redox Reactions

Oxidation Reduction Worksheet Answers Free Read Oxidation Reduction Reaction Worksh In 2020 Scientific Notation Word Problems Worksheet Template Pre Algebra Worksheets

Tang 02 Balancing Redox Reactions 2 Redox Reactions Chemistry Lessons Chemistry Education

Tang 02 Balancing Redox Reactions 2 Redox Reactions Organic Reactions Reactions

Qualified Oxidation Reduction Reactions Worksheet Kimia

Pin On Customize Printable Worksheet Templates

Tang 02 Balancing Redox Reactions 2 Lezioni Di Scienze Chimica Scienza

Redox Reactions Redox Reactions Chemistry Worksheets Chemistry Education

Tang 02 Balancing Redox Reactions Redox Reactions Teaching Chemistry Teaching Science

Oxidation Reduction Worksheet Answers 25 Redox Review Worksheet Answers In 2020 Scientific Notation Word Problems Word Problem Worksheets Pre Algebra Worksheets

Redox Reactions Exercise With Solutions Redox Reactions Chemical Equation Reactions

Picture Redox Reactions Free Math Worksheets Chemistry Notes

Balancing Chemical Equations 10th Higher Ed Worksheet Chemical Equation Balancing Equations Chemistry Worksheets


