Alkaline conditions then we have to put in an extra step to balance the equation. 5 co i 2 o 5 5 co 2 i 2 in basic solution.

Picture Redox Reactions Free Math Worksheets Chemistry Notes
4e 4h o 2 2h 2 o.

Balancing redox reactions worksheet with answers. Balancing oxygen and hydrogen in basic redox reactions sometimes can be difficult because both oh and h 2 o contain both elements. If the redox reaction was carried out in basic solution i e. Balancing redox reactions worksheet 1 balance each redox reaction in.
H 2o 2 cr 2o 7 2 o 2 cr 3 9. Mno 2 mn 2o 3 balance each redox reaction in acid solution using the half reaction method. Balancing redox reactions in basic solution.
So 4 2 so 2 7. Write balanced equations for the following redox reactions. Which equation represents the half reaction that takes place at.
A trick to get around this is to balance any troublesome half reaction or the entire redox reaction first as if it were in acid using h and h 2 o. Write balanced equations for the following reactions. Worksheet 5 balancing redox reactions in acid and basic solution balance each half reaction in basic solution.
No no 3 6. Atoms other than o and h then o then h. Cl2 2e 2cl c.
If you re seeing this message it means we re having trouble loading external resources on our website. In the reaction mg cl2 mgcl2 the correct half reaction for the oxidation that occurs is a. Questions pertaining to redox reactions.
Identify the pair of elements undergoing oxidation and reduction by checking oxidation states. Balance each of the following half cell reactions. Teo 3 2 n 2o 4 te no 3 10.
2 nabr cl 2 2 nacl br 2 b. Cr 2o 7 2 cr3 5. Divide the skeleton reaction into two half reactions each of which contains the oxidized and reduced forms of one of the species 2.
Cr oh 3 br 2 cro 4 2 br in basic solution 10 oh 2 cr oh 3 3 br 2 2 cro 4 2 8 h 2 o. In each case assume that the reaction takes place in an acidic solution also state whether the reaction is oxidation or reduction. You cannot have electrons appear in the final answer of a redox reaction.
The steps for balancing redox reactions in basic solution are. If you re behind a web filter. Balance the atoms and charges in each half reaction atoms are balanced in order.
Balancing redox reactions in base. You can in a half reaction but remember half reactions do not occur alone they occur in reduction oxidation pairs 2 here are the correct half reactions. The reaction that takes place in a chemical cell is best classi ed as a.
And that is wrong because there is an electron in the final answer. Balancing redox equations method 2. Fe 2 o 3 3 co 2 fe 3 co 2 in acidic solution c.
Mg mg2 2e d. Ws 4 balancing redox reactions. Mn 2 bio3 æ mno4 bi 3 mno4 s2o3 2 æ s4o6 2 mn 2.
Questions pertaining to redox reactions.

Tang 02 Balancing Redox Reactions 2 Lezioni Di Scienze Chimica Scienza

Tang 02 Balancing Redox Reactions 2 Redox Reactions Chemistry Notes Reactions

Pin On Customize Printable Worksheet Templates

49 Balancing Equations Practice Worksheet Answers In 2020 Balancing Equations Chemical Equation Equations

An In Depth Introduction To Balancing Redox Equations Complete With A Step By Step Guide To Make Sure Nothing Is Misse Redox Reactions Mcat Mcat Study Schedule

Oxidation Reduction Worksheet Answers Free Read Oxidation Reduction Reaction Worksh In 2020 Scientific Notation Word Problems Worksheet Template Pre Algebra Worksheets

Tang 02 Balancing Redox Reactions Redox Reactions Teaching Chemistry Teaching Science

Redox Reactions Redox Reactions Chemistry Worksheets Chemistry Education

Tang 02 Balancing Redox Reactions 2 Redox Reactions Chemistry Notes Reactions
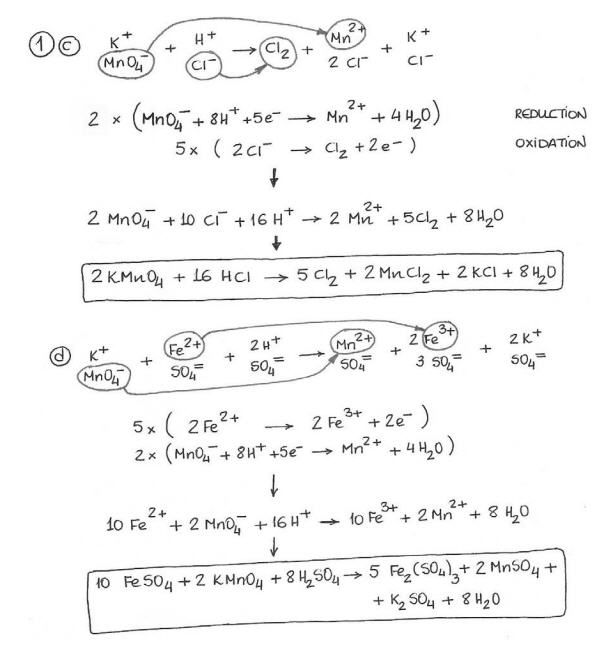
Pin By Saitech Informatics On Balancing Of Chemical Reaction Equations Distance Time Graphs Worksheets Redox Reactions

Balancing Equations Worksheet Answers Beautiful As Chemistry Balancing Redox Equations Worksheet In 2020 Balancing Equations Persuasive Writing Prompts Equations

Chemistry Multiple Multiple Choice Types Of Chemical Reactions Chemistry Chemical Reactions Multiple Choice

Tang 02 Balancing Redox Reactions 2 Redox Reactions Organic Reactions Reactions

Redox Reactions Exercise With Solutions

Chapter 6 Oxidation Reduction Redox Reactions Teaching Chemistry Chemistry Notes

Balancing Chemical Equations Worksheets With Answers In 2020 Balancing Equations Chemical Equation Equations

Tang 02 Balancing Redox Reactions 2 Redox Reactions Chemistry Lessons Chemistry Education

Redox Balancing Redox Reactions Chemistry Lagrange College

Oxidation Reduction Redox Reactions Balancing Redox Reactions Chemistry Net Redox Reactions Teaching Chemistry Chemistry Lessons

