If the redox reaction was carried out in basic solution i e. The latter is easier to use with redox reactions in aqueous solution and if necessary can be adapted to many situations that are not in aqueous solution.

Pin On Customize Printable Worksheet Templates
Worksheet 5 balancing redox reactions in acid and basic solution balance each half reaction in basic solution.

Balancing redox reactions worksheet with solutions. Fe 2 o 3 co fe co 2 in acidic solution c. You cannot have electrons appear in the final answer of a redox reaction. Alkaline conditions then we have to put in an extra step to balance the equation.
5 co i 2 o 5 5 co 2 i 2 in basic solution. Write balance equations for the following redox reactions. Teo 3 2 n 2o 4 te no 3 10.
Balancing redox reactions in basic solution. B balance the oxygen atoms with h 2 o. O 2 sb h 2 o 2 sbo 2 in basic solution hint.
2 nabr cl 2 2 nacl br 2 b. Cr 2o 7 2 cr3 5. Cr oh 3 br 2 cro 4 2 br in basic solution.
Often students lose hope and struggle to solve it. Identify the pair of elements undergoing oxidation and reduction by checking oxidation states. Fe 2 o 3 3 co 2 fe 3 co 2 in acidic solution c.
Balance the atoms in each half reaction a balance all other atoms except h and o. Write balanced equations for the following reactions. The steps for balancing redox reactions in basic solution are.
H 2o 2 cr 2o 7 2 o 2 cr 3 9. Write balanced equations for the following redox reactions. So 4 2 so 2 7.
Convert the following redox reactions to the ionic form. Mno 2 mn 2o 3 balance each redox reaction in acid solution using the half reaction method. No no 3 6.
B identify and write out all redox couples in reaction. Cr oh 3 br 2 cro 4 2 br in basic solution 10 oh 2 cr oh 3 3 br 2 2 cro 4 2 8 h 2 o. Mn 2 bio3 æ mno4 bi 3 mno4 s2o3 2 æ s4o6 2 mn 2.
H2s kmno4 k2so4 mns h2o s. Balance redox equations using the ion electron method in an acidic solutions. Nabr cl 2 nacl br 2 b.
There are two principal methods for balancing redox equations. Balancing redox reactions in acidic solution problems 1 10 fifteen examples problems 26 50 balancing in basic solution. Co i 2 o 5 co 2 i 2 in basic solution hint.
Balancing redox equations by the ion electron method. If you are struggling as well then all you need balancing equations worksheet with answers. You can in a half reaction but remember half reactions do not occur alone they occur in reduction oxidation pairs.
Balancing requires a lot of practice knowledge of reactions formulae valances symbols and techniques. Balancing redox reactions worksheet 1 balance each redox reaction in. C balance the hydrogen atoms with h d in a basic medium add one oh to each side for every h step 4.
Write balanced equations for the following reactions. C combine these redox couples into two half reactions.

Tang 02 Balancing Redox Reactions 2 Redox Reactions Organic Reactions Reactions

Tang 02 Balancing Redox Reactions 2 Lezioni Di Scienze Chimica Scienza

Picture Redox Reactions Free Math Worksheets Chemistry Notes

Balancing Equations Worksheet Answers Beautiful As Chemistry Balancing Redox Equations Worksheet In 2020 Balancing Equations Persuasive Writing Prompts Equations

Oxidation Reduction Worksheet Answers Free Read Oxidation Reduction Reaction Worksh In 2020 Scientific Notation Word Problems Worksheet Template Pre Algebra Worksheets

Redox Reactions Exercise With Solutions

Tang 02 Balancing Redox Reactions Redox Reactions Teaching Chemistry Teaching Science

Types Of Chemical Reactions Worksheet 19 Sample Balancing Chemical Equations Wor In 2020 Chemistry Worksheets Sight Word Worksheets Kindergarten Subtraction Worksheets

Redox Balancing Redox Reactions Chemistry Lagrange College

Tang 02 Balancing Redox Reactions 2 Redox Reactions Chemistry Notes Reactions

Tang 02 Balancing Redox Reactions 2 Redox Reactions Chemistry Lessons Chemistry Education

Ncert Solutions For Class 11 Chemistry Chapter 8 Redox Reactions 025 Redox Reactions 11th Chemistry Chemistry

Redox Reaction Redox Reactions Teaching Chemistry Chemistry Notes
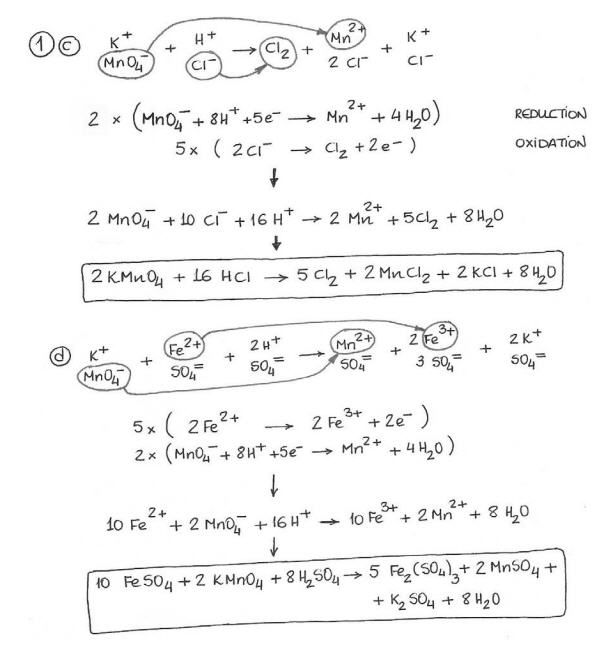
Pin By Saitech Informatics On Balancing Of Chemical Reaction Equations Distance Time Graphs Worksheets Redox Reactions

Ncert Solutions For Class 11 Chemistry Chapter 8 Redox Reactions 055

Balancing Redox Reactions Half Reaction Method Redox Reactions Reactions Chemistry

Qualified Oxidation Reduction Reactions Worksheet Kimia

Chemistry Multiple Multiple Choice Types Of Chemical Reactions Chemistry Chemical Reactions Multiple Choice

Redox Reactions Redox Reactions Chemistry Worksheets Chemistry Education

