Periodic table coloring worksheet. What is the atomic mass of mercury.
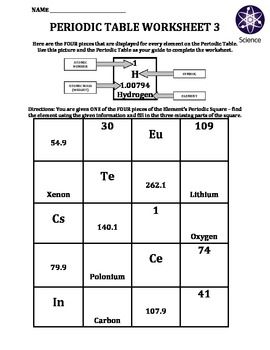
Worksheet Periodic Table Worksheet 3 Science Teaching Resources Chemistry Classroom Teaching Chemistry
The periodic table worksheet this worksheet is to be used to go along with the corresponding reading.

Periodic table worksheet. List the symbols for two transition metals. Similarities in reactivity between elements 5. Name the place.
Periodic table packet 1 name period directions. You will learn the not only find the atomic number mass and symbol but you will learn how to translate that to a a number of protons neutrons and electrons for a stable atom. Ni is the symbol for what element.
Names of groups in the periodic table 4. Using the periodic table of elements sheet 1 use the chart and your knowledge of chemistry to complete the blanks for atoms of each element. The table is useful not only for students in high school or college taking their chemistry classes.
How group number relates to outer shell electrons 3. Periodic table worksheets know your periodic table soup to nuts with our periodic table worksheets. The periodic table worksheet with answers 1 the meaning of group number and how this relates to reactivity trends 2.
Find the names of cities using the numbers of the elements. What is the atomic symbol for silver. A tabular display of chemical elements the periodic table helps students feel at home with chemical elements and keeps them thoroughly and immaculately informed of the atomic numbers electronic configurations and chemical properties of elements.
Differences between elements and. Using the numbers given find the elements and put them together to make the names. The element that has the atomic number 17 is.
Use your periodic table to complete the worksheet. Use the following table and legend to answer the questions to the best of your ability. Legend solid liquid gas synthetic alkali metals alkali earth metals transition metals rare earth metals.
Using the numbers given find the elements and put them together to make the names of places. Importance of the periodic table. Periodic table questions sheet.
It also helps scientists in predicting all types of chemical reactions happening inside an element.

Science Matter Periodic Table Worksheet With Key Matter Science Chemistry Worksheets Teaching Science

Worksheet Blank Periodic Table Persuasive Writing Prompts Physical Science Middle School Science Worksheets

The Tterry23 Shop On Teachersnotebook Com Physical Science High School Chemistry Worksheets Science Teaching Resources

Periodic Chart Jpg 535 693 Pixels Chemistry Classroom Science Chemistry High School Science

Worksheet Periodic Table Worksheet 1 Chemistry Worksheets Teaching Chemistry Chemistry Lessons

Periodic Table Worksheet Key Chemistry Worksheets Printable Worksheets Worksheet Template

Clock Buddies Worksheet Flag President Periodic Table Buddies And More Chemistry Worksheets Chemistry Classroom Science Worksheets

Organizing The Periodic Table Worksheet Chemistry Classroom Chemistry Lessons Teaching Chemistry
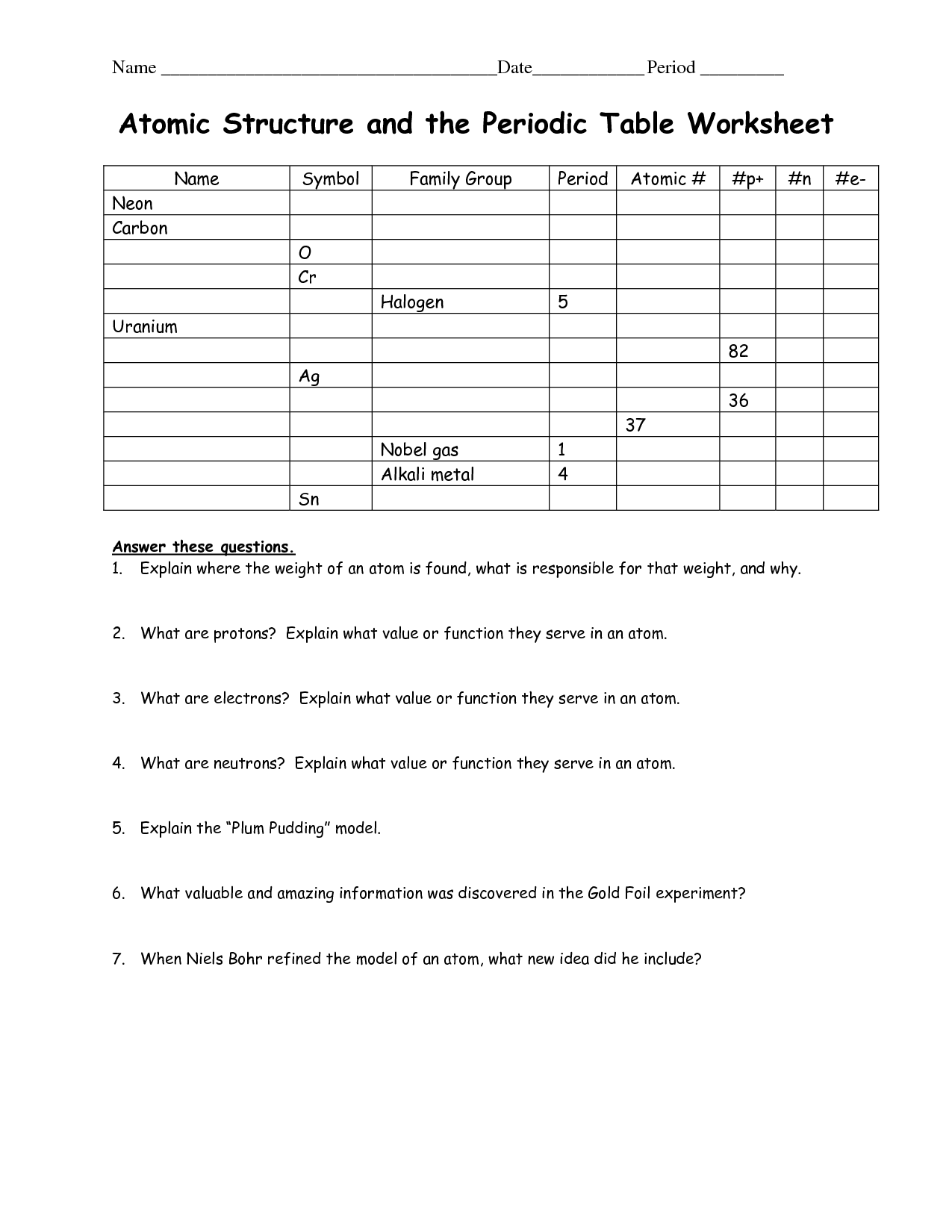
Atomic Structure Worksheet And Periodic Table Chemistry Worksheets Atomic Structure Chemistry

Worksheet Periodic Table Worksheet 3 Science Worksheets Chemistry Classroom 8th Grade Science

Periodic Table Worksheets Chemistry Worksheets Chemistry Classroom Teaching Chemistry

All Worksheets Free Periodic Table Worksheets Periodic Table Facts Worksheet Answers Deli Chemistry Worksheets Periodic Table Chemistry Periodic Table

Interpreting The Periodic Table Activity Page Chemistry Worksheets Teaching Chemistry Chemistry Lessons

Organizing The Periodic Table Worksheet Chemistry Classroom Chemistry Lessons Teaching Chemistry

Electron Shells Periodic Table Worksheet Chemistry Worksheets Chemistry Classroom Teaching Chemistry

Periodic Table Worksheet Education Com Periodic Table Learning Science Science Worksheets

Periodic Table Basics Worksheet Answer Key Chemistry Worksheets Chemistry Classroom Teaching Chemistry

Free Periodic Table Worksheet Chemistry Worksheets Chemistry Classroom Teaching Chemistry

Print The Periodic Table Worksheets And Use A Periodic Table To Find Missing Element Information And Lear Worksheet Template Worksheets Word Problem Worksheets

