The steps for balancing redox reactions in basic solution are. Identify the pair of elements undergoing oxidation and reduction by checking oxidation states.
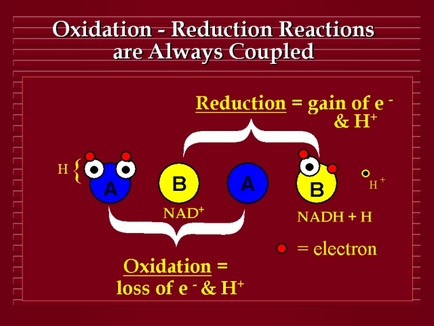
Lesson 3 Predicting Redox Reactions Grade12uchemistry
Al agno 3 18.

Spontaneity of redox reactions worksheet answers. The reaction that takes place in a chemical cell is best classi ed as a. H 2o 2 cr 2o 7 2 o 2 cr 3 9. Balancing redox reactions in basic solution.
If the redox reaction was carried out in basic solution i e. Mg mg2 2e d. So 4 2 so 2 7.
Prediction according to nelson text all single replacement reactions are spontaneous. If the answer is no write a balanced equation for the reaction that would occur. A strip of magnesium metal is placed in an aqueous 1 mol l 1 copper ii sulfate solution.
Mn 2 bio3 æ mno4 bi 3 mno4 s2o3 2 æ s4o6 2 mn 2. What is the question asking you to do. Mno 2 mn 2o 3 balance each redox reaction in acid solution using the half reaction method.
Teo 3 2 n 2o 4 te no 3 10. Balance the redox equation using the half reaction method. Worksheet 5 balancing redox reactions in acid and basic solution balance each half reaction in basic solution.
A voltaic cell uses a. Lab 13 2 spontaneity of redox reactions purpose the purpose of this lab is to test the assumption that all single replacement reactions are spontaneous. No no 3 6.
Cr 2o 7 2 cr3 5. Circle each formula that is able to lose an electron o. Worksheet 3 spontaneous and non spontaneous redox reactions.
Spontaneity of redox reactions under standard state conditions calculate δg and k for the following reaction at 25 c. 3mg s 2al3 aq 3mg2 aq 2al s use the equation below to calculate k. Will a spontaneous redox reaction occur.
Can you keep 1 m hno. Based on the stopgops approach to problem solving. Cl2 2e 2cl c.
E o for the redox reaction not given. Can you keep 1 m hcl in an ag container. Quiz worksheet spontaneous redox reactions in electrolytic cells quiz.
Alkaline conditions then we have to put in an extra step to balance the equation. In the reaction mg cl2 mgcl2 the correct half reaction for the oxidation that occurs is a. Problem which combinations of cu s pb s ag s zn s and their aqueous metal ion solutions produce spontaneous reactions.
Balancing redox reactions worksheet 1 balance each redox reaction in. Choose an answer and hit next. Determine if the redox reaction as given is spontaneous.
Which equation represents the half reaction that takes place at. You will receive your score and answers at the end.

Redox Bundle Practice Worksheets Practices Worksheets Number Worksheets Worksheets
Oxidation Reduction Reactions Redox

Section 9 3 Predicting Redox Reactions

Image Result For Gibbs Free Energy And Reduction Potential Free Energy Redox Reactions Reduction Potential
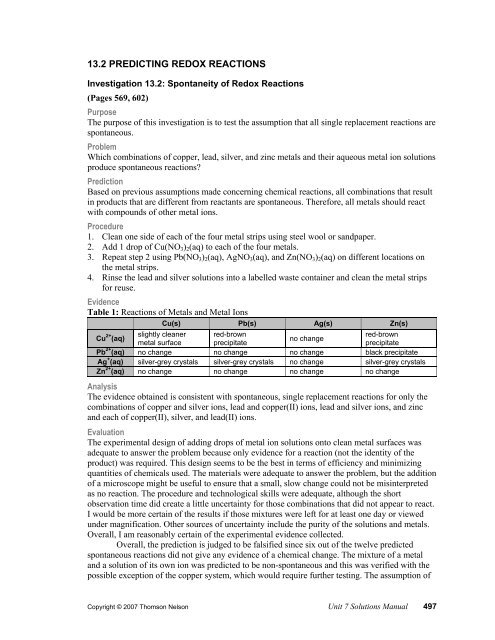
13 2 Predicting Redox Reactions
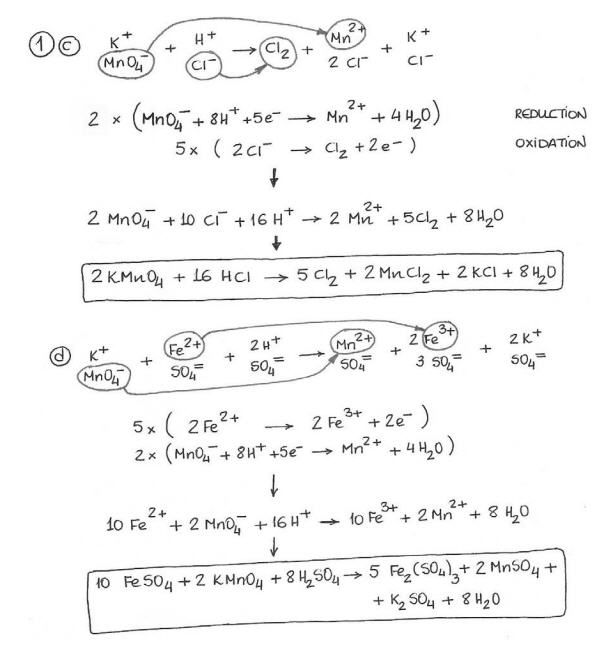
Pin By Saitech Informatics On Balancing Of Chemical Reaction Equations Distance Time Graphs Worksheets Redox Reactions

Imageedit 4 6384949106 Jpg Electrochemistry Galvanic Cell Chemistry Classroom

Balancing Redox Reactions In Acidic And Basic Conditions Youtube

Spontaneity And Redox Reactions Video Khan Academy

Chemical Reactions Types Worksheet Unique 16 Best Of Types Chemical Reactions Worksheets Chessmu In 2020 Reaction Types Persuasive Writing Prompts Chemical Reactions

Electrochemistry Featuring Electrolysis And Fuel Cells Teaching Chemistry Science Chemistry Electrochemistry

Tang 02 Balancing Redox Reactions 2 Redox Reactions Chemistry Lessons Chemistry Education

Click To Download Redox Rules Posters For Vce Chemistry Teaching Chemistry Chemistry Ap Chemistry

Voltaic Cells Chemistry Chemistry Lesson Plans Electrochemistry

How To Assign Oxidation Numbers In 2020 Reducing Agent Oxidation Redox Reactions

Tang 02 Balancing Redox Reactions 2 Redox Reactions Organic Reactions Reactions

Redox Reactions Notes And Galvanic Cells Galvanic Cell Chemistry Notes Redox Reactions